 There he was increasingly occupied with studies of artificial disintegration of atomic nuclei-changes in nuclear structure induced by high-energy particulate streams. Rutherford remained at Manchester until 1919, when he succeeded Thomson as the head of the Cavendish Laboratory at Cambridge. Presuming radioactive sources of solar and terrestrial energy opened the way for the revival of Darwin's theory of natural selection by the 1920s. Rutherford himself estimated as early as 1904 that the significant amount of heat generated by radioactivity in the earth's crust would extend the earth's age much longer than previously believed. It provided a means to surmount what had been one of the most imposing barriers to acceptance of Charles Darwin's evolutionary theory-that with the sun and earth presumed cooling over time, the earth could not sustain temperatures supporting life for a time long enough to have produced all the changes evident in the fossil record via Darwinian evolution. Moreover, his recognition that the energy involved in radioactive (nuclear) transformations far exceeded energy produced in chemical reactions had ramifications beyond physical science. His findings furnished impressive evidence that atoms exist and helped resolve a long-standing tension in physical science between the concepts of atom and element by tying elemental identity to the constitution of the atomic nucleus. He had determined what radioactivity was and had revealed essential aspects of the atom's structure. /rutherford-atomic-model-141483941-5c3bb44bc9e77c0001c7a15d.jpg)
The impacts of Rutherford's work up to 1914 were enormous. Rutherford's conclusion: atoms in the foil must be mostly empty space, with a tiny volume occupied by a massive and highly charged "nucleus." By 1913, combined speculations of Rutherford and the Dane Niels Bohr (1885–1962) had identified the nucleus as: (1) the positively charged component of an atom whose total charge determines the atom's elemental identity, and (2) the site of radioactivity (the origin of alpha, beta, and gamma emissions). By 1911 this work indicated that one out of eight thousand alpha particles was deflected ninety degrees or greater by the very thin foil. Puzzled by effects seeming to indicate that air molecules slightly diverted alpha particles from their original directions, he embarked on a systematic investigation of this "scattering" by orienting alpha particle streams at very thin metal foils and determining how interaction with atoms in the foil affected the direction of the alpha particles' motions. Alpha, Rutherford speculated, was a stream of positively charged helium ions.īy 1908, a year after moving to the University of Manchester in England, Rutherford was exploiting the alpha emission in a new experimental direction. Gamma appeared to be high-energy electromagnetic radiation. Beta seemed clearly to be fast-moving electrons (the subatomic, negatively charged particles that Thomson had identified in 1896 in studies of electrical effects in gases at very low pressures). By now recognizing that three major components could constitute these emissions-alpha, beta, and gamma-he was dedicated to identifying them. In the following year, Rutherford hypothesized that radioactive emission occurs at the instant that an atom changes its elemental identity. By 1902 they were convinced that radioactivity involved transmutations of atoms of one element into atoms of another element. He and the chemist Frederick Soddy undertook monumental studies of radioactivity, combining chemical analyses with investigations of ionization effects. In 1898, Rutherford was appointed professor of physics at McGill University in Montreal, Canada. Soon turning to investigations of radioactivity, which Antoine- Henri Becquerel (1852–1908) had just discovered, Rutherford applied the technique of quantitatively assessing ionization effects in air surrounding radioactive substances-he was able to identify two components of emissions from radioactive substances: alpha was easily absorbed by a thin foil, while beta penetrated the foil. He and Thomson studied the ability of X-rays (recently discovered by the German Wilhelm Conrad Roentgen) to "ionize" gases into positively and negatively charged particles and thus increase the ability of the gases to conduct electricity. degree at Canterbury College, Christchurch, New Zealand. 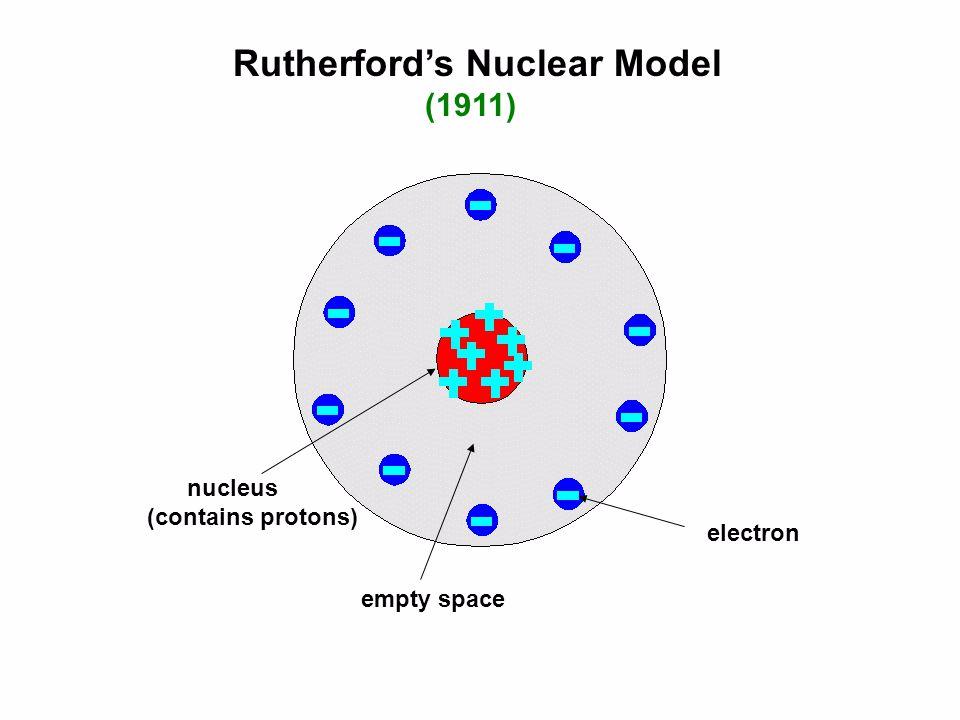 
Born in New Zealand, Rutherford came to the Cavendish Laboratory at Cambridge University, England, in 1896 to study with Joseph John Thomson shortly after earning the M.A. RUTHERFORD, ERNEST (1871–1937), New Zealand-born scientist.Įrnest Rutherford's experimental work was essential to early-twentieth-century revolutionary developments in atomic physics. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
